
Phone: 86-10-62782861
Email: wenwenzeng @ tsinghua.edu.cn

The Laboratory of Wenwen Zeng
The research interest in the Zeng lab is to explore the brain-body interaction underlying energy balance and diseases to better understand fundamental biological processes and benefit therapeutic intervention. Currently, our major direction is energy balance, including metabolic homeostasis and diseases, neural and immune regulation of metabolism and neuroinflammation. In depth understanding of these processes may provide key insights into the therapeutics of metabolic related diseases, including obesity, diabetes, and immunological disorders.

Wenwen Zeng, Ph.D.
Professional Experience
1998 - 2002 B.S., Biological Sciences, Tsinghua University, China
2003 - 2009 Ph.D., The University of Texas Southwestern Medical Center, USA (Mentor: Zhijian "James" Chen, Ph.D.)
2009 - 2011 Postdoctoral Research Fellow, Department of Discovery Immunology, Genentech Inc., USA (Mentor: Harinder Singh, Ph.D.)
2011 - 2014 Postdoctoral Fellow, Howard Hughes Medical Institute, Laboratory of Molecular Genetics, The Rockefeller University, USA (Mentor: Jeffrey M. Friedman, Ph.D.)
2014 - present Principal Investigator, School of Medicine & Institute for Immunology, Tsinghua, University, China
2018 - present Associate director, Beijing Key Lab for Immunological Research on Chronic Diseases
2020 - 2023 Associate Professor (Tenured), School of Medicine & Institute for Immunology, Tsinghua, University, China
2023 - present Professor (Tenured), School of Medicine & Institute for Immunology, Tsinghua, University, China
Recognitions and Awards
2022 The National Science Fund for Distinguished Young Scholar
2018 The National Science Fund for Excellent Young Scholar
2018 Young Investigator Award, Lipid Metabolism and Bioenergetics Subsociety of the Biophysical Society of China
2016 Outstanding Young Scholar Award, Qiu-Shi Science & Technologies Foundation
2015 National Young Investigator Award
Academic Activities
2020 -present Editorial board member, Oxford Open Immunology
Editorial Experience
2023 – present, Editorial board member, Journal of Genetics and Genomics
2022 – present, Editorial board member, Advanced Science
2022 – present, Scientific Editor, Fundamental Research
2022 – present, Associate Editor, AJP Endocrinology and Metabolism
2020 – present, Editorial board member, Oxford Open Immunology
2021 – Guest Associate Editor for Multiple Sclerosis and Neuroimmunology,
Frontiers in Immunology, Frontiers in Neurology
Post-Docs
Xiaofan Ding

I obtained my Ph.D. degree in biology from Tsinghua University in 2020, focusing on neuroimmune regulation of tissue homeostasis. Currently, I’m a postdoctoral fellow in Zeng Lab and study inflammation and diseases.
Graduate Students
Xinmin Qian

Xinmin Qian, Ph.D candidate starting from 2017, takes interest in basketball and running but is sadly suffering from multiple collateral damage. He is an amiable and loving person who is crazy about all the animals, especially his model organism--mus musculus. The main research direction is exploring mechanisms of intra-adipose sympathetic plasticity.
Wenran Ren

I gained my bachelor degree from Nankai University. My study mainly focuses on the crosstalk between sympathetic nervous system and metabolism.
Nan Deng

Nan Deng is a Ph.D student (from 2018) in the School of Medicine. She attended Tsinghua University for her bachelor's degree. Currently, her research focuses on neuroimmune response
Meng Jiang

I graduated from Beijing Normal University with my bachelor degree. My study mainly focuses on the antibody production.
Yixin Dai

Yixin attended Nankai University for her bachelor's degree and studied bioscience. During her studies she became interested in research, which prompted her to apply for a doctoral degree in Tsinghua University. She joined Zeng lab in 2019. Currently, her research focuses on neuron-immune cell crosstalk.
Mengxue Sun

Mengxue Sun, Ph.D student (from 2019) in School of Medicine, Bachelor’s degree in biotechnology from University of Science and Technology of China; research focus: neural-immune metabolism.
Qingqing Li

Ph.D student (from 2019) in School of Medicine, got bachelor degree from Shandong University. My research interest is focused on neuroimmune response.
Haoyu Yuan

Haoyu Yuan, Ph.D student (from 2019) in School of Medicine, Bachelor’s degree from Tsinghua University; research focus: neuro-immune crosstalk.
Jianhui Chen

Jianhui Chen comes from Fujian, china. He got his bachelor's degree in Tsinghua university and studied life science. He joined Zeng lab in 2020. Currently, he mainly investigates the sympathetic activity and inflammation.
Yongwen Wan

Ph.D. student (from 2020) in Tsinghua University. Yongwen attended Ocean University of China (OUC) for his bachelor's degree and joined Zeng lab in 2020. His research mainly focuses on the neural regulation of metabolism and immunometabolism.
Meng Hua

Meng is a Ph.D. candidate from 2021. She obtained her bachelor’s degree from China Agricultural University and Cornell University. Her research interest lies in neuroimmune crosstalk.
Weijia Chen

Weijia Chen, Ph.D. student (from 2022) in Tsinghua University, got his bachelor's degree in Zhejiang University and studied life science. His research mainly focuses on neuroimmune response.
Jian Song

Jian Song, Ph.D candidate starting from 2022, bachelor's degree from Northeast Forestry University. He looks serious but has a warm heart. He loves doing calligraphy and playing ping-pong. And about him, the interesting fact is that the one who stares at him for a long time will be paid a shy smile. His current research is focused on neuroimmune response.
Lab Alumni
Haochen Jiang:Institute of Systems Biology, Jianghan University
Zhongsheng Hu:Nanjing Legend Biotechnology Co., Ltd.
Energy balance
Efferent signals from the central nervous system represent a key layer of regulation of white adipose tissue (WAT). We exploit the volume fluorescence-imaging technique to visualize the neural arborizations in mouse inguinal WAT at single-fiber resolution. The imaging reveals a dense network of sympathetic arborizations that had been previously undetected by conventional methods, with sympathetic fibers being in close apposition to > 90% of adipocytes. We demonstrate that these sympathetic fibers originate from the celiac ganglia, which are activated by cold challenge. Sympathetic-specific deletion of TrkA receptor or pharmacologic ablation by 6-hydroxydopamine abolishes these intra-adipose arborizations and, as a result, cold-induced beiging of inguinal WAT. Furthermore, we find that local sympathetic arborizations function through beta-adrenergic receptors in this beiging process. These findings uncover an essential link connecting efferent neural signals with metabolism of individual adipocytes.
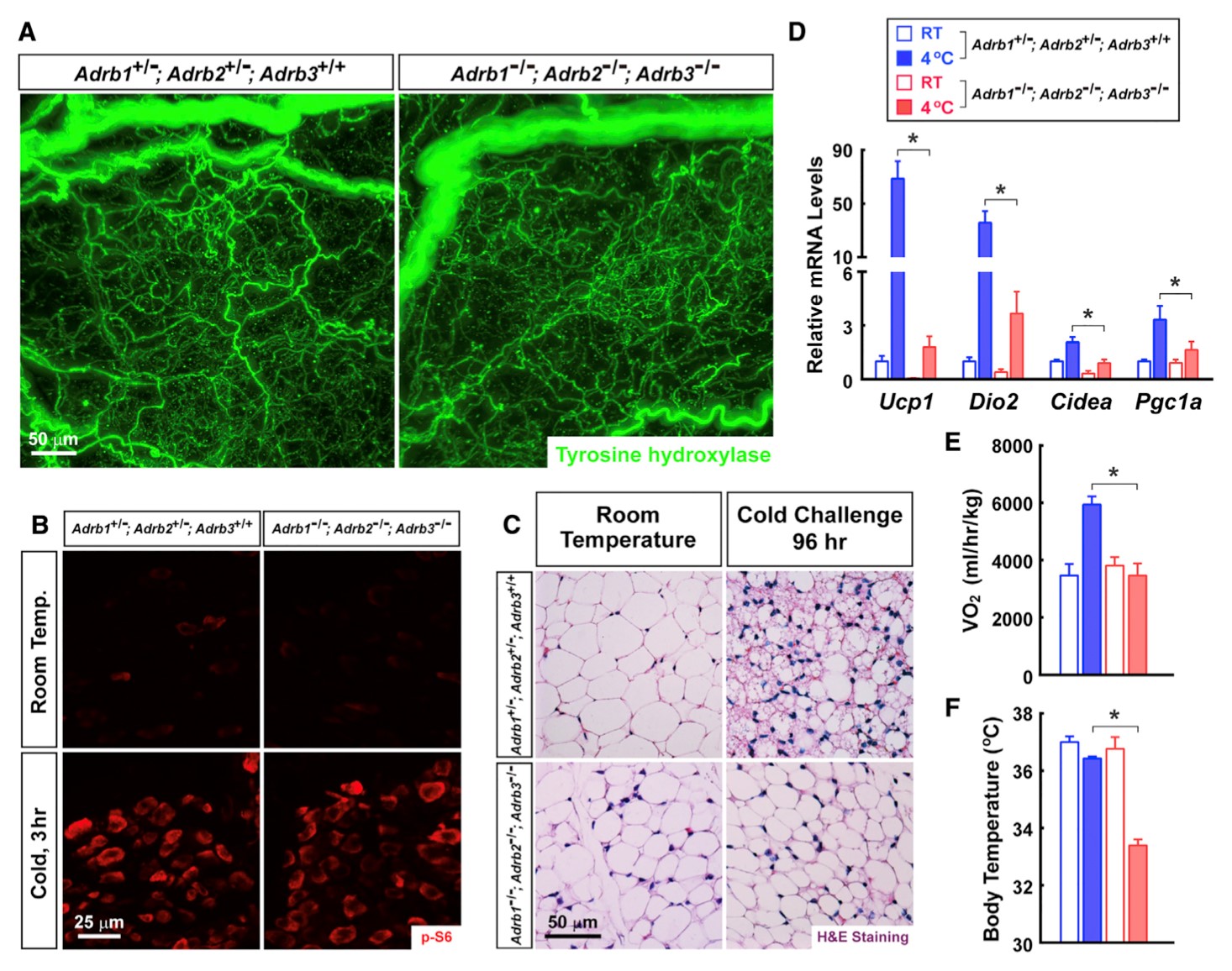
Jiang H, Ding X, Cao Y, Wang H, Zeng W. Dense Intra-adipose Sympathetic Arborizations Are Essential for Cold-Induced Beiging of Mouse White Adipose Tissue. Cell Metab. 2017 Oct 3;26(4):686-692.e3. doi: 10.1016/j.cmet.2017.08.016.
Neuroinflammation
Neuroinflammation is a crucial mechanism in many neurological disorders. Injury to the peripheral sensory nerves leads to a neuroinflammatory response in the somatosensory pathway, from dorsal root ganglia (DRG) to the spinal cord, contributing to neuropathic pain. We find that ciliary neurotrophic factor (CNTF), highly expressed in Schwann cells, mediates neuroinflammatory response through the activating signal transducer and activator of transcription 3 (STAT3) and inducing interleukin 6 (IL-6) in sensory neurons. Cntf deficiency attenuates neuroinflammation in DRG and the spinal cord with alleviated pain post-injury. Recombinant CNTF applied to the sensory nerves recapitulates neuroinflammation in the DRG and spinal cord, with consequent pain development. We delineate the CNTF-STAT3-IL-6 axis in mediating the onset and progression of the inflammatory cascade from the periphery to the spinal cord with therapeutic implications for neuropathic pain.
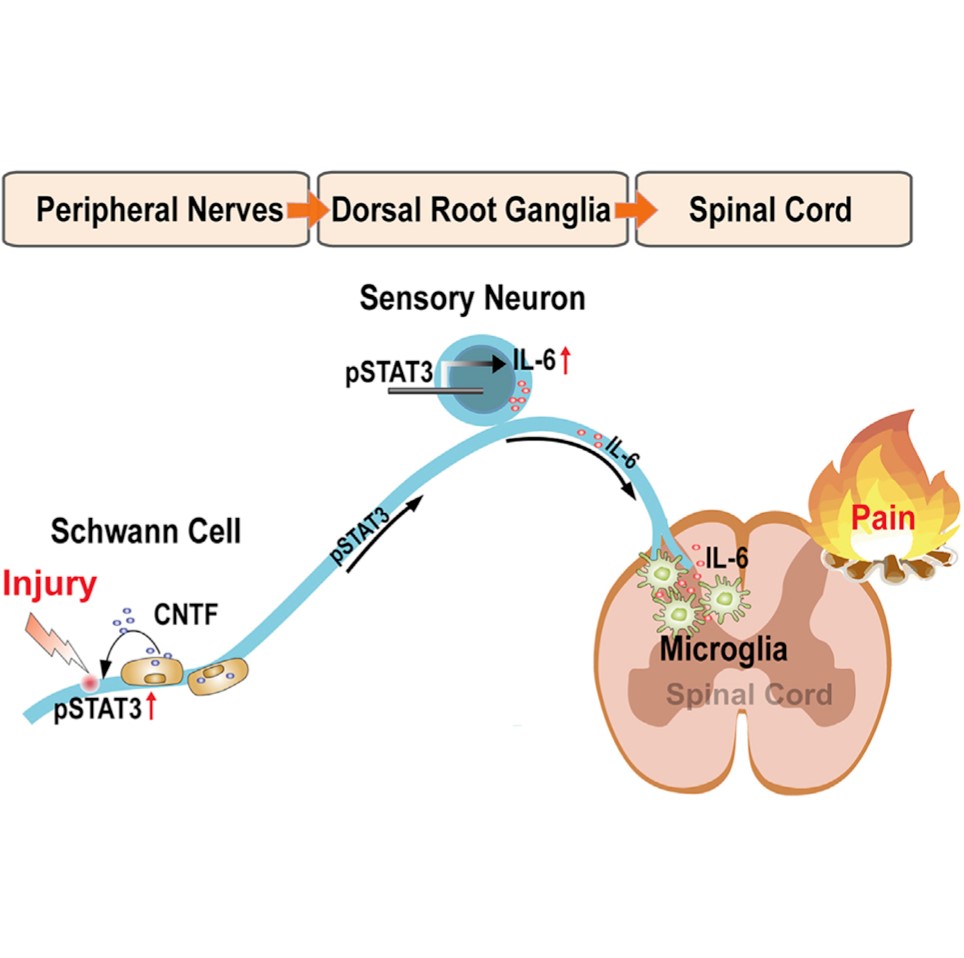
Hu Z, Deng N, Liu K, Zhou N, Sun Y, Zeng W. CNTF-STAT3-IL-6 Axis Mediates Neuroinflammatory Cascade across Schwann Cell-Neuron-Microglia. Cell Rep. 2020 May 19;31(7):107657. doi: 10.1016/j.celrep.2020.107657.
We find that dual leucine zipper kinase (DLK) is required for the neuronal intrinsic immune response to induce cytokines and chemokines such as Ccl2, Ccl7, and Ccl12 upon nerve injury. The DLK-controlled injury response in sensory neurons could regulate CD11b+ immune cell infiltration in the dorsal root ganglia, as well as microgliosis and astrogliosis in the spinal dorsal horn but not the ventral horn. Deficiency of Dlk drastically alleviates the neuropathic pain elicited by chronic constriction injury of the sciatic nerve. Thus, DLK is an essential component that mediates the neuronal intrinsic immune response to nerve injury in sensory neurons and regulates inflammation in the spinal cord.
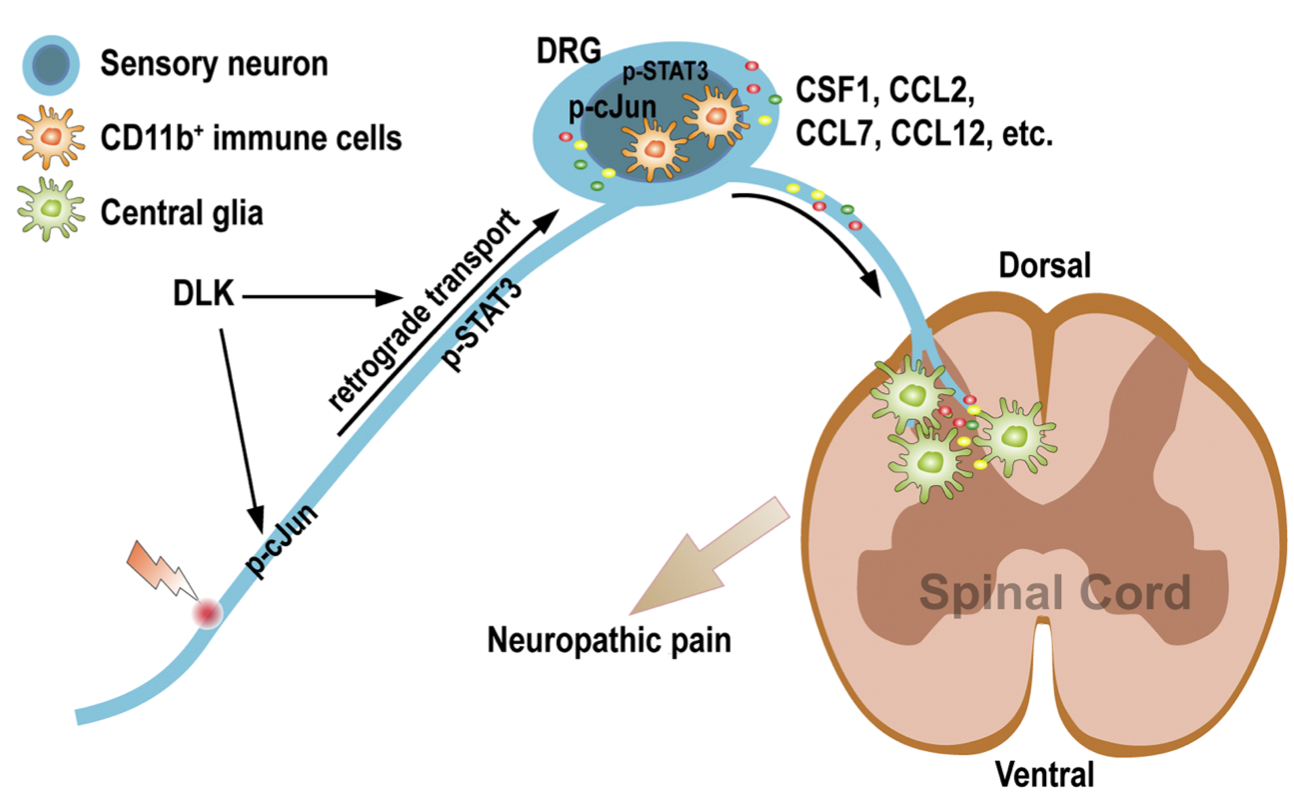
Hu Z, Deng N, Liu K, Zeng W. DLK mediates the neuronal intrinsic immune response and regulates glial reaction and neuropathic pain. Exp Neurol. 2019 Dec;322:113056. doi: 10.1016/j.expneurol.2019.113056. Epub 2019 Sep 5.
ORCID: 0000-0001-8544-3318
ResearcherID: D-7276-2017
Selected Publications (#corresponding author and lead author, *equal contribution)
1. Meng, X., Chen J. #, and Zeng, W.# (2022) Stromal cell-derived NGF controls sympathetic innervation in subcutaneous fat. JLR.
2. Ding, X., Chen J., and Zeng, W.# (2022) Neuroimmune regulation in the pancreas. Fundamental Research.
3. Guo, Z., Yang, H., Zhang, J.R., Zeng, W.#, and Hu, X.# (2022) Leptin receptor signaling sustains metabolic fitness of alveolar macrophages to attenuate pulmonary inflammation. Sci Adv 8, eabo3064. 10.1126/sciadv.abo3064.
4. Zeng, W.#, Yang, F.#, Shen, W.#, Zhan, C.#, Zheng, P.#, and Hu, J. (2022) Interactions between central nervous system and peripheral metabolic organs, Science China Life Sciences. doi: 10.1007/s11427-021-2103-5.
5. Xie, H., Heier, C., Meng, X., Bakiri, L., Pototschnig, I., Tang, Z., Schauer, S., Baumgartner, V.J., Grabner, G.F., Schabbauer, G., Wolinski, H., Robertsong, G.R., Hoefler, G., Zeng, W., Wagner, E.F., Schweiger, M.#, and Zechner, R.# (2022) An Immune-Sympathetic Neuron Communication Axis Guides Adipose Tissue Browning in Cancer-associated Cachexia, Proc Natl Acad Sci U S A. 119.
6. Meng, X.*, Qian, X.*, Ding, X.*, Wang, W.*, Yin, X.#, Zhuang, G.#, and Zeng, W.# (2022) Eosinophils regulate intra-adipose axonal plasticity, Proc Natl Acad Sci U S A. 119.
7. Zeng, W.#, and Qian, X. (2021) Neuronal Regulation of Adipose Tissue Biology in Neuron Signaling in Metabolic Regulation (Tong, Q., ed), CRC Press.
8. Qian, X., Meng, X., Zhang, S., and Zeng, W.# (2021) Neuroimmune regulation of white adipose tissues, FEBS J.
9. Meng, X., and Zeng, W.# (2021) Fat mesenchyme closes the neural-ILC2 circuit, Immunity. 54, 2191-2193.
10. Zhuo, W.#, Zhou, H., Guo, R., Yi, J., Zhang, L., Yu, L., Sui, Y., Zeng, W., Wang, P.#, and Yang, M.# (2021) Structure of intact human MCU supercomplex with the auxiliary MICU subunits, Protein Cell. 12, 220-229.
11. Zhang, S., Zhou, J., Zhang, Y., Liu, T., Friedel, P., Zhuo, W., Somasekharan, S., Roy, K., Zhang, L., Liu, Y., Meng, X., Deng, H., Zeng, W., Li, G.#, Forbush, B.#, and Yang, M.# (2021) The structural basis of function and regulation of neuronal cotransporters NKCC1 and KCC2, Commun Biol. 4, 226.
12. Sui, Y., and Zeng, W.# (2020) MS4A4A Regulates Arginase 1 Induction during Macrophage Polarization and Lung Inflammation in Mice, Eur J Immunol. 50, 1602-1605.
13. Meng, X., Zeng, W., Young, S. G.#, and Fong, L. G. (2020) GPIHBP1, a partner protein for lipoprotein lipase, is expressed only in capillary endothelial cells, J Lipid Res. 61, 591.
14. Hu, Z.*, Deng, N.*, Liu, K., Zhou, N., Sun, Y., and Zeng, W.# (2020) CNTF-STAT3-IL-6 Axis Mediates Neuroinflammatory Cascade across Schwann Cell-Neuron-Microglia, Cell Rep. 31, 107657.
15. Hu, Z., Deng, N., Liu, K., and Zeng, W.# (2019) DLK mediates the neuronal intrinsic immune response and regulates glial reaction and neuropathic pain, Exp Neurol. 322, 113056.
16. Wang, Q.*, Zhang, S.*, Liu, T.*, Wang, H., Liu, K., Wang, Q., and Zeng, W.# (2018) Sarm1/Myd88-5 Regulates Neuronal Intrinsic Immune Response to Traumatic Axonal Injuries, Cell Rep. 23, 716-724.
17. Hu, Z., and Zeng, W.# (2018). Splenic Leukocytes Get the Nerves up for Myelopoiesis. Immunity 49, 1-3.
18. Zhang, S.*, Li, N.*, Zeng, W., Gao, N.#, and Yang, M.# (2017). Cryo-EM structures of the mammalian endo-lysosomal TRPML1 channel elucidate the combined regulation mechanism. Protein Cell 8, 834-847.
19. Jiang, H.*, Ding, X.*, Cao, Y., Wang, H., and Zeng, W.# (2017). Dense Intra-adipose Sympathetic Arborizations Are Essential for Cold-Induced Beiging of Mouse White Adipose Tissue. Cell Metab 26, 686-692 e683.
20. Zeng, W.*, Pirzgalska, R.M.*, Pereira, M.M., Kubasova, N., Barateiro, A., Seixas, E., Lu, Y.H., Kozlova, A., Voss, H., Martins, G.G., et al. (2015). Sympathetic neuro-adipose connections mediate leptin-driven lipolysis. Cell 163, 84-94.
21. Zeng, W.*, Lu, Y.H.*, Lee, J., and Friedman, J.M. # (2015). Reanalysis of parabiosis of obesity mutants in the age of leptin. Proc Natl Acad Sci U S A 112, E3874-3882.
22. Peng, Y.*, Zeng, W.*, Ye, H., Han, K.H., Dharmarajan, V., Novick, S., Wilson, I.A., Griffin, P.R., Friedman, J.M., and Lerner, R.A.# (2015). A General Method for Insertion of Functional Proteins within Proteins via Combinatorial Selection of Permissive Junctions. Chem Biol 22, 1134-1143.
23. Glasmacher, E.*, Agrawal, S.*, Chang, A.B.*, Murphy, T.L.*, Zeng, W.*, Vander Lugt, B., Khan, A.A., Ciofani, M., Spooner, C.J., Rutz, S., et al. (2012). A genomic regulatory element that directs assembly and function of immune-specific AP-1-IRF complexes. Science 338, 975-980.
24. Rutz, S., Noubade, R., Eidenschenk, C., Ota, N., Zeng, W., Zheng, Y., Hackney, J., Ding, J., Singh, H., and Ouyang, W.# (2011). Transcription factor c-Maf mediates the TGF-beta-dependent suppression of IL-22 production in T(H)17 cells. Nat Immunol 12, 1238-1245.
25. Zeng, W.*, Sun, L.*, Jiang, X., Chen, X., Hou, F., Adhikari, A., Xu, M., and Chen, Z.J.# (2010). Reconstitution of the RIG-I pathway reveals a signaling role of unanchored polyubiquitin chains in innate immunity. Cell 141, 315-330.
26. Zeng, W., Xu, M., Liu, S., Sun, L.#, and Chen, Z.J.# (2009). Key role of Ubc5 and lysine-63 polyubiquitination in viral activation of IRF3. Mol Cell 36, 315-325.
27. Xu, M., Skaug, B., Zeng, W., and Chen, Z.J.# (2009). A ubiquitin replacement strategy in human cells reveals distinct mechanisms of IKK activation by TNFalpha and IL-1beta. Mol Cell 36, 302-314.
28. Xia, Z.P., Sun, L., Chen, X., Pineda, G., Jiang, X., Adhikari, A., Zeng, W., and Chen, Z.J.# (2009). Direct activation of protein kinases by unanchored polyubiquitin chains. Nature 461, 114-119.
29. Zeng, W., and Chen, Z.J.# (2008). MITAgating viral infection. Immunity 29, 513-515.
30. Yang, M., Xu, G., Li, S., Sun, L., Shi, N., Zeng, W., Pang, H., Zhang, W., and Rao, Z.# (2003). Crystallization and preliminary crystallographic analysis of the extracellular fragment of FcalphaRI/CD89. Acta Crystallogr D Biol Crystallogr 59, 2251-2253.
31. Sun, Z.H., Lai, Y.L., Zeng, W., Zhao, D., Zuo, H.C., and Xie, Z.P.# (2003). Neural stem/progenitor cells survive and differentiate better in PD rats than in normal rats. Acta Neurochir Suppl 87, 169-174.
32. Sun, Z.H., Lai, Y.L., Zeng, W., Zhao, D., Ye, Z.W., Zuo, H.C., and Xie, Z.P.# (2003). Mesencephalic progenitors can improve rotational behavior and reconstruct nigrostriatal pathway in PD rats. Acta Neurochir Suppl 87, 175-180.








Copyright © 2017 Institute for Immunology Tsinghua University
Contact Address: Room D302, Medical Science Building, Tsinghua University, Beijing 100084, China
Tel: (86) 10-62776420 Fax: (86) 10-62776420
